For a complete list of publications, please click here
The many roles of myosins in filopodia, microvilli and stereocilia.
Houdusse A & Titus MA (2021) Curr Biol 31:R586-R602.
doi: 10.1016/j.cub.2021.04.005
Filopodia, microvilli and stereocilia represent an important group of plasma membrane protrusions. These specialized projections are supported by parallel bundles of actin filaments and have critical roles in sensing the external environment, increasing cell surface area, and acting as mechanosensors. This review highlights recent progress in understanding the diverse roles myosins play in filopodia, microvilli and stereocilia.
VASP mediated actin dynamics activate and recruit a filopodia myosin
Arthur AL, Crawford A, Houdusse A & Titus MA (2021) Elife 10:e68082.
doi: 10.7554/eLife.68082.
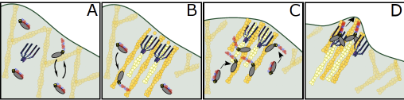
The actin polymerase VASP and a MyTH-FERM (MF) myosin, DdMyo7 in amoeba, are essential for filopodia initiation. The work here shows DdMyo7 is highly localized the actin rich-leading edge of the cell. Myosin recruitment and activation in Dictyostelium requires a local cortical actin network generated by VASP and together they shape the actin network to promote extension of parallel bundles of actin during filopodia formation. Thus, filopodia initiation requires close collaboration between an actin binding protein, the state of the actin cytoskeleton and MF myosin activity.
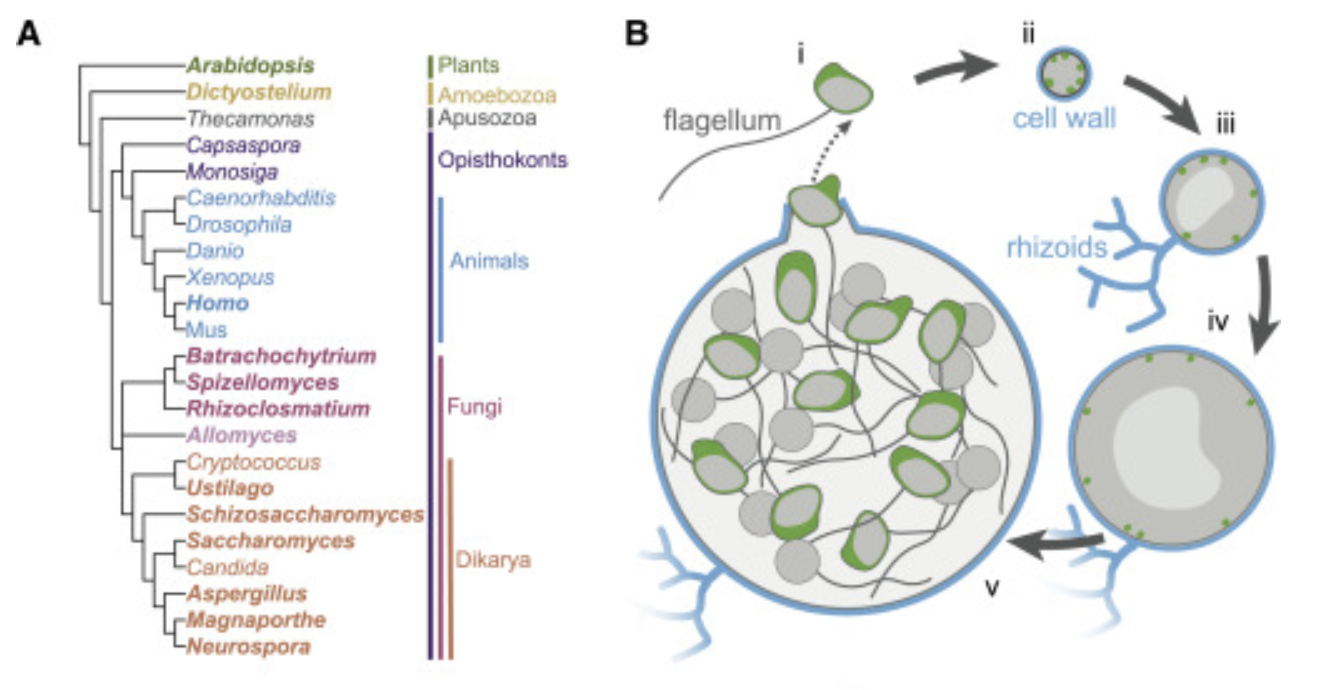
The actin networks of chytrid fungi reveal evolutionary loss of cytoskeletal complexity in the fungal kingdom.
Prostak SM, Robinson K, Titus MA & Fritz-Laylin LK (2021) Curr. Biol 31:1192-1205.e6. Link
Cells from across the eukaryotic tree use actin polymer networks for a wide variety of functions, including endocytosis, cytokinesis, and cell migration. Chytrid fungi provide a unique opportunity to study actin cytoskeletal evolution. The presence of animal- and yeast-like actin cytoskeletal components in the genome combined with the intermediate actin phenotypes in Bd suggests that the simplicity of the yeast cytoskeleton may be due to evolutionary loss.
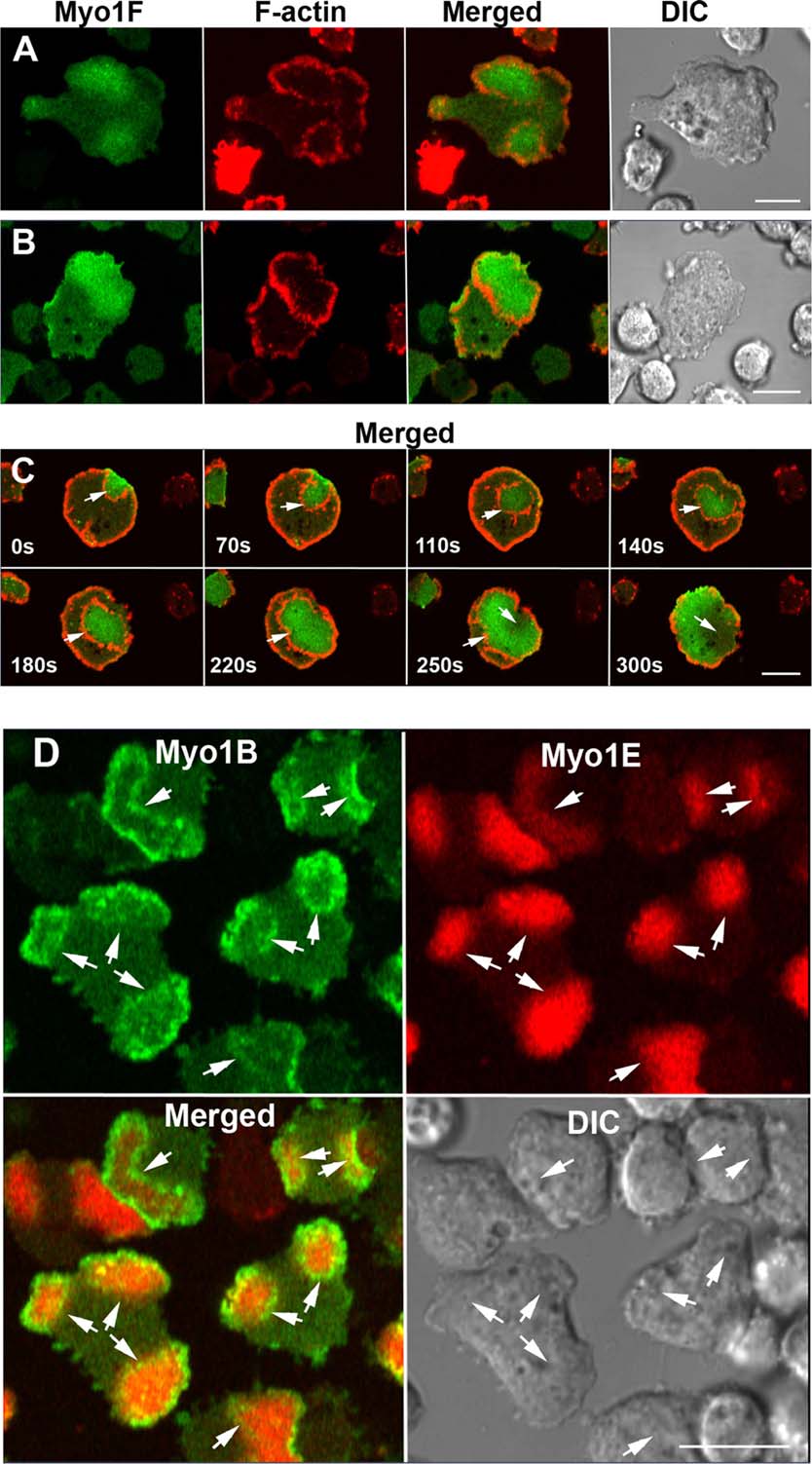
Dictyostelium myosin 1F and myosin 1E inhibit actin waves in a lipid-binding-dependent and motor-independent manner.
Brzeska H*, Bagnoli M, Korn ED§ & Titus MA§ (2020) Cytoskeleton 77:295-302.
*corresponding author § equal contributors Link
Actin waves are F‐actin‐rich entities traveling on the ventral plasma membrane by the treadmilling mechanism. Localization of five Dictyostelium Class I myosins synchronizes with localization and propagation of actin waves. Ectopic expression of the PIP3 specific short‐tail Myo1F myosin inhibits wave formation, short‐tail Myo1E has similar but weaker inhibitory effect, but long‐tail Myo1D does not affect waves. The results suggest that PIP3 specificity and the presence of two membrane‐binding sites are required for inhibition of actin waves, and that inhibition may be caused by crosslinking of PIP3 heads groups.
Basic-hydrophobic (BH) sites are localized in conserved positions inside and outside of PH domains and affect localization of Dictyostelium myosin 1s
Brzeska H*, Gonzalez J, Korn ED§ & Titus MA§ (2020) Mol. Biol. Cell 31:101-117
*corresponding author § equal contributors Link
Myosin 1s have critical roles in linking membranes to the actin cytoskeleton via direct binding to acidic lipids. Lipid binding may occur through PIP3/PIP2-specific PH domains or nonspecific ionic interactions involving basic-hydrophobic (BH) sites but the mechanism of myosin 1s distinctive lipid targeting is poorly understood. Mutations of BH sites affect localization of all Dictyostelium myosin 1s reveraling that these sites are important determinants of motor targeting and may have a similar role in the localization of other myosin 1s.
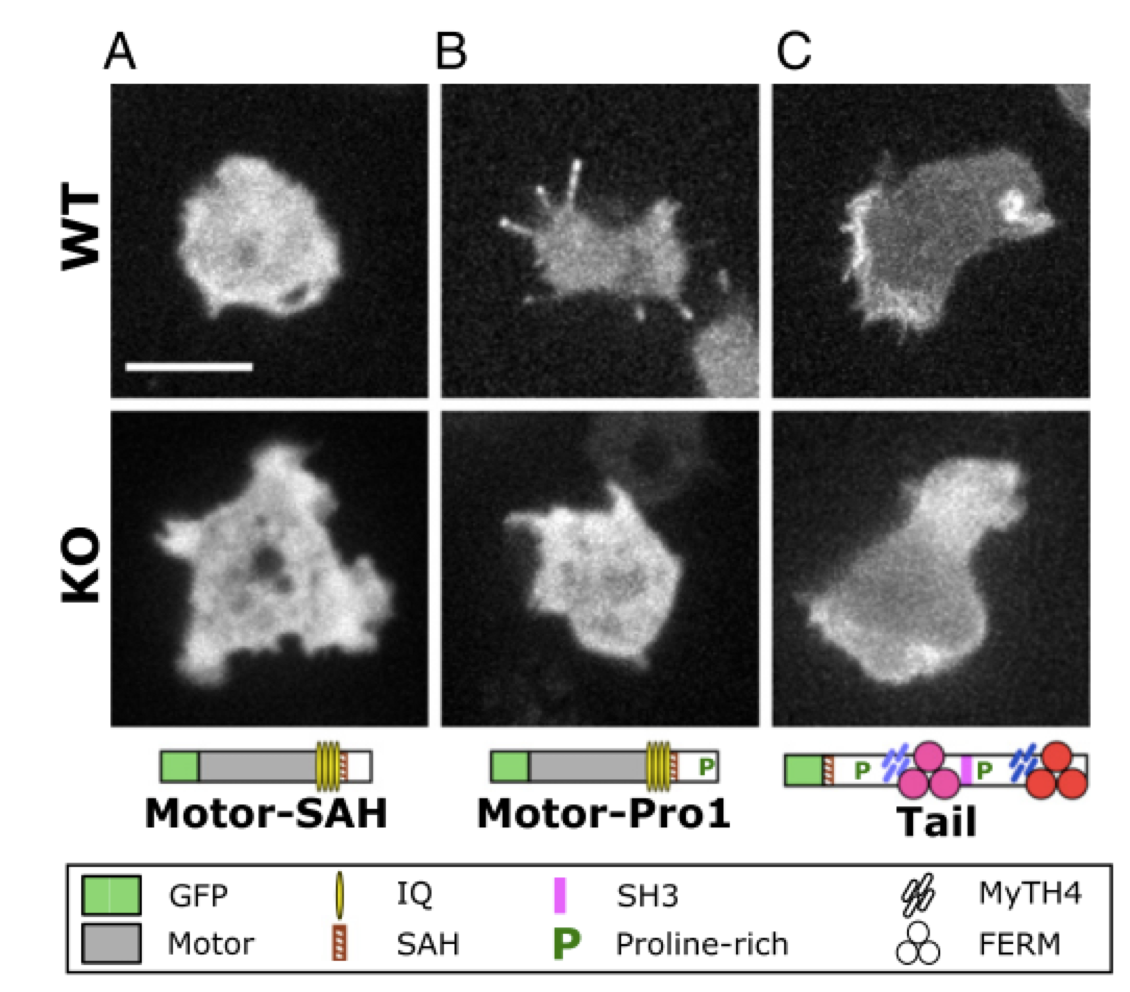
Optimized filopodia formation requires myosin tail domain cooperation
Arthur, AL., Songster, LD., Sirkia, H, Bhattacharya, A, Kikuti, C, Borrega, FP, Houdusse, A, Titus, MA. (2019) PNAS 116 (44): 22196–204 PMID:31611382. Link
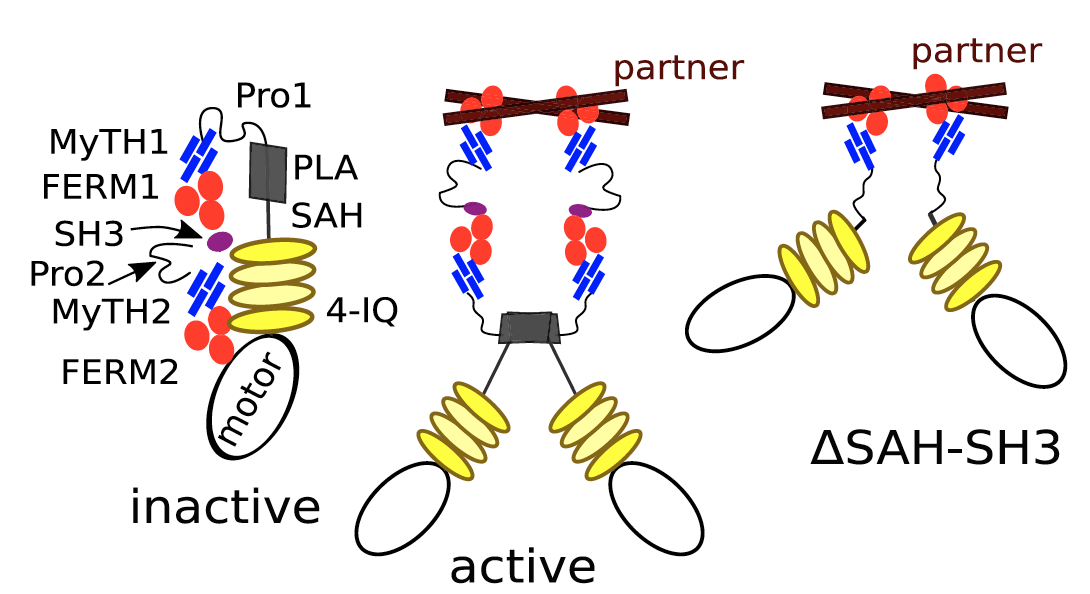
Filopodia formation in Amoebozoa and Metazoa requires the phylogenetically diverse MyTH4-FERM (MF) myosins DdMyo7 and Myo10, respectively. While Myo10 is known to form antiparallel dimers, DdMyo7 lacks a coiled-coil domain in its proximal tail region. The DdMyo7 lever arm is found to play a role in both autoinhibition and function while the proximal tail region can mediate weak dimerization, and likely works in cooperation with the C-terminal MF domain to promote partner-mediated dimerization. Thus, the principles of MF myosin-based filopodia formation are conserved via divergent mechanisms for dimerization.
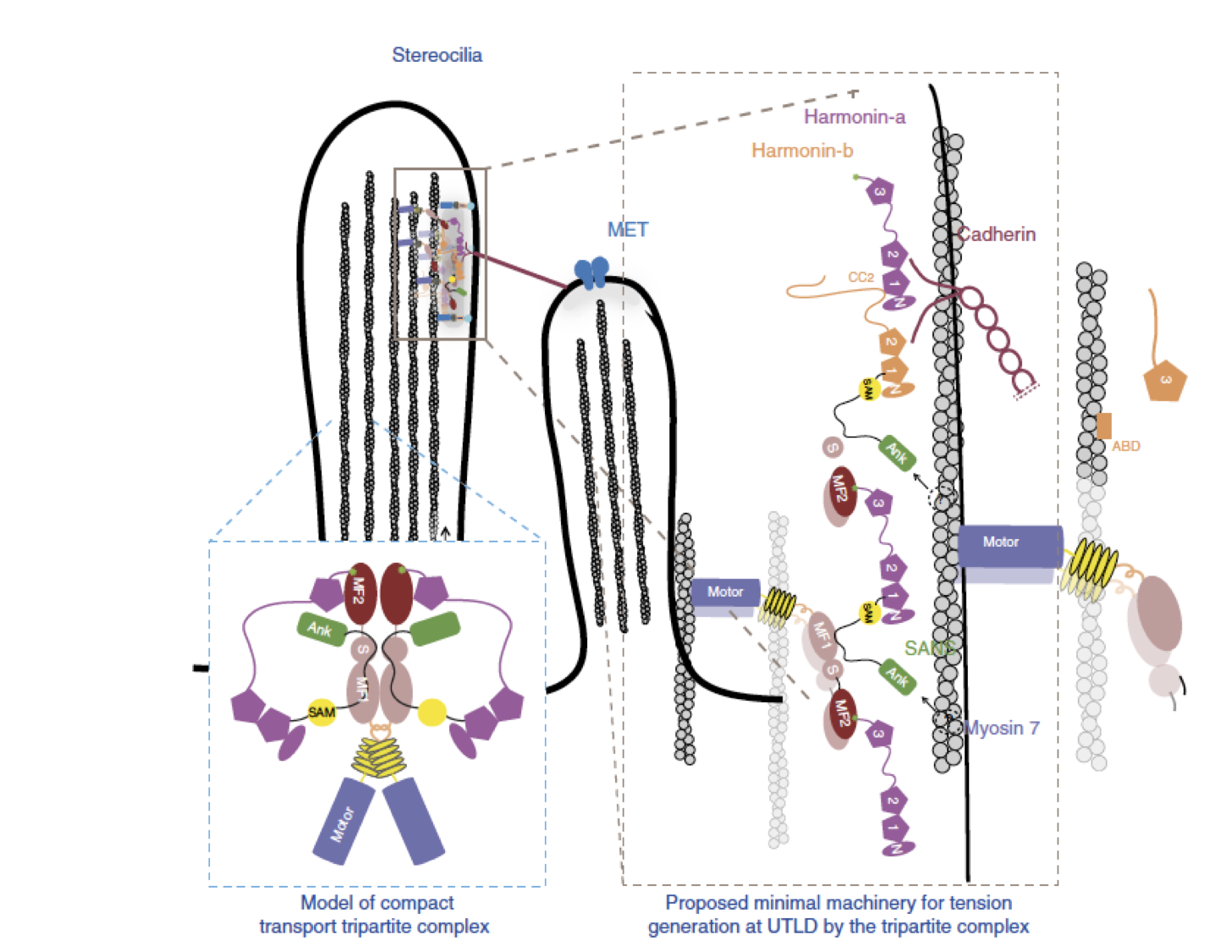
Myosin 7 and its adaptors link cadherins to actin.
Yu IM, Planelles-Herrero VJ, Sourigues Y, Moussaoui D, Sirkia H, Kikuti C, Stroebel D, Titus MA*, Houdusse A* (2017) Nature Comm. 8:15864. * co-senior authors PDF Link
Cadherin linkages between adjacent stereocilia and microvilli are essential for mechanotransduction and maintaining their organization. They are anchored to actin through interaction of their cytoplasmic domains with related tripartite complexes consisting of a class VII myosin and adaptor proteins: Myo7a/SANS/Harmonin in stereocilia and Myo7b/ANKS4B/Harmonin in microvilli. Here, we determine high-resolution structures of Myo7a and Myo7b C-terminal MyTH4-FERM domain (MF2) and unveil how they recognize harmonin using a novel binding mode. Systematic definition of interactions between domains of the tripartite complex elucidates how the complex assembles and prevents possible self-association of harmonin-a. Several Myo7a deafness mutants that map to the surface of MF2 disrupt harmonin binding, revealing the molecular basis for how they impact the formation of the tripartite complex and disrupt mechanotransduction. Our results also suggest how switching between different harmonin isoforms can regulate the formation of networks with Myo7a motors and coordinate force sensing in stereocilia.
MyTH4-FERM myosins have an ancient and conserved role in filopod formation
Petersen, KJ, Goodson, HV, Arthur, AL, Luxton, GWG, Houdusse, A & Titus, MA (2016) PNAS 113:E8059-E8068. PDF Link
The formation of filopodia in Metazoa and Amoebozoa requires the activity of myosin 10 (Myo10) in mammalian cells and of Dictyostelium unconventional myosin 7 (DdMyo7) in the social amoeba Dictyostelium. However, the exact roles of these MyTH4-FERM myosins (myosin tail homology 4-band 4.1, ezrin, radixin, moesin; MF) in the initiation and elongation of filopodia are not well defined and may reflect conserved functions among phylogenetically diverse MF myosins. Phylogenetic analysis of MF myosin domains suggests that a single ancestral MF myosin existed with a structure similar to DdMyo7, which has two MF domains, and that subsequent duplications in the metazoan lineage produced its functional homolog Myo10. The essential functional features of the DdMyo7 myosin were identified using quantitative live-cell imaging to characterize the ability of various mutants to rescue filopod formation in myo7-null cells. The two MF domains were found to function redundantly in filopod formation with the C-terminal FERM domain regulating both the number of filopodia and their elongation velocity. DdMyo7 mutants consisting solely of the motor plus a single MyTH4 domain were found to be capable of rescuing the formation of filopodia, establishing the minimal elements necessary for the function of this myosin. Interestingly, a chimeric myosin with the Myo10 MF domain fused to the DdMyo7 motor also was capable of rescuing filopod formation in the myo7-null mutant, supporting fundamental functional conservation between these two distant myosins. Together, these findings reveal that MF myosins have an ancient and conserved role in filopod formation.
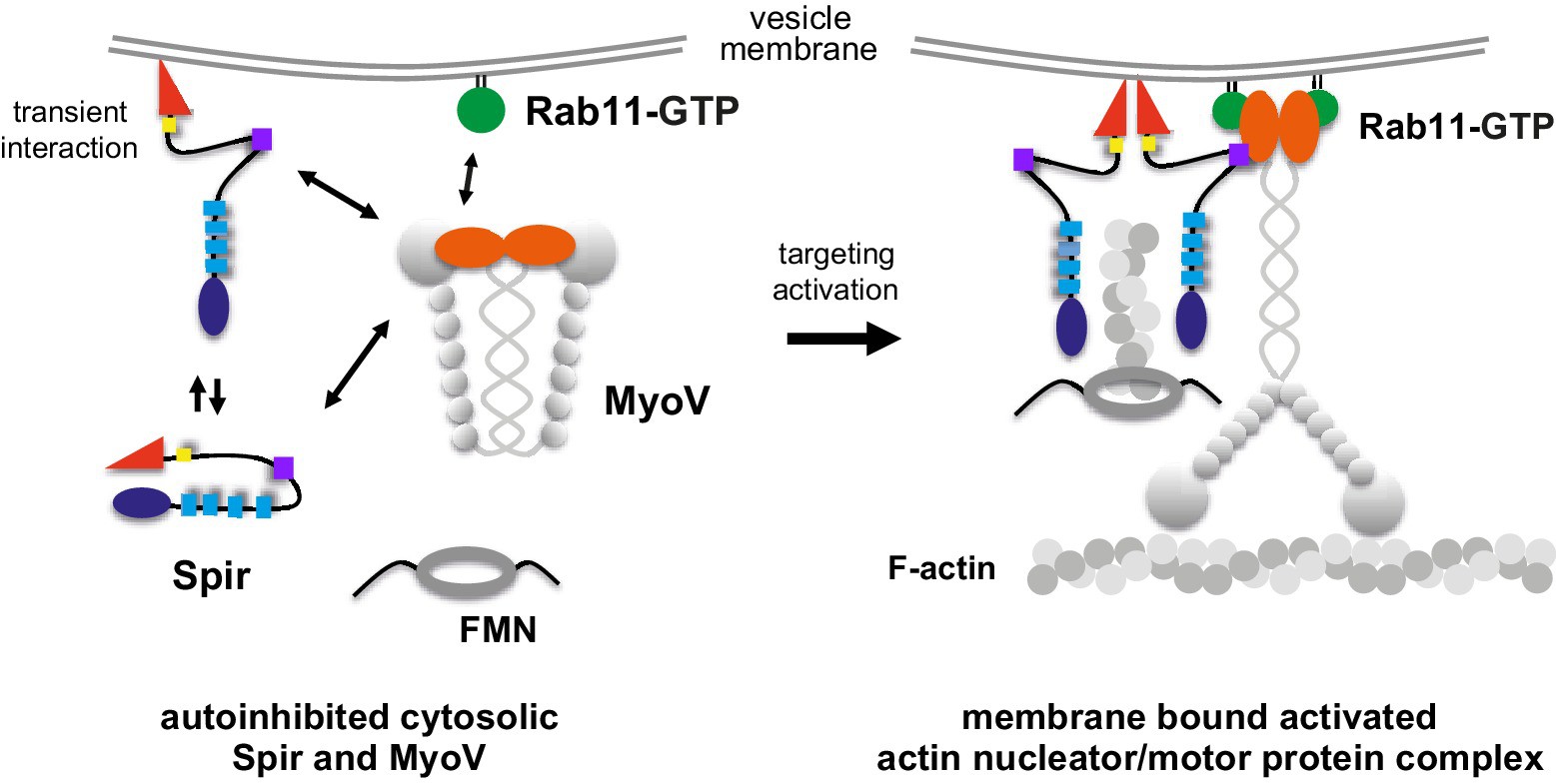
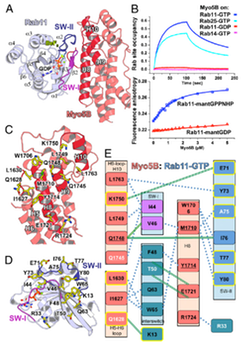
Coordinated recruitment of Spir actin nucleators and myosin V motors to Rab11 vesicle membranes
Pylypenko, O. et al. (2016) Coordinated recruitment of spir actin nucleators and myosin V motors to rab11 vesicle membranes. eLife 5:e17523. PDF Link
Abstract:
There is growing evidence for a coupling of actin assembly and myosin motor activity in cells. However, mechanisms for recruitment of actin nucleators and motors on specific membrane compartments remain unclear. Here we report how Spir actin nucleators and myosin V motors coordinate their specific membrane recruitment. The myosin V globular tail domain (MyoV-GTD) interacts directly with an evolutionarily conserved Spir sequence motif. We determined crystal structures of MyoVa-GTD bound either to the Spir-2 motif or to Rab11 and show that a Spir-2:MyoVa:Rab11 complex can form. The ternary complex architecture explains how Rab11 vesicles support coordinated F-actin nucleation and myosin force generation for vesicle transport and tethering. New insights are also provided into how myosin activation can be coupled with the generation of actin tracks. Since MyoV binds several Rab GTPases, synchronized nucleator and motor targeting could provide a common mechanism to control force generation and motility in different cellular processes.
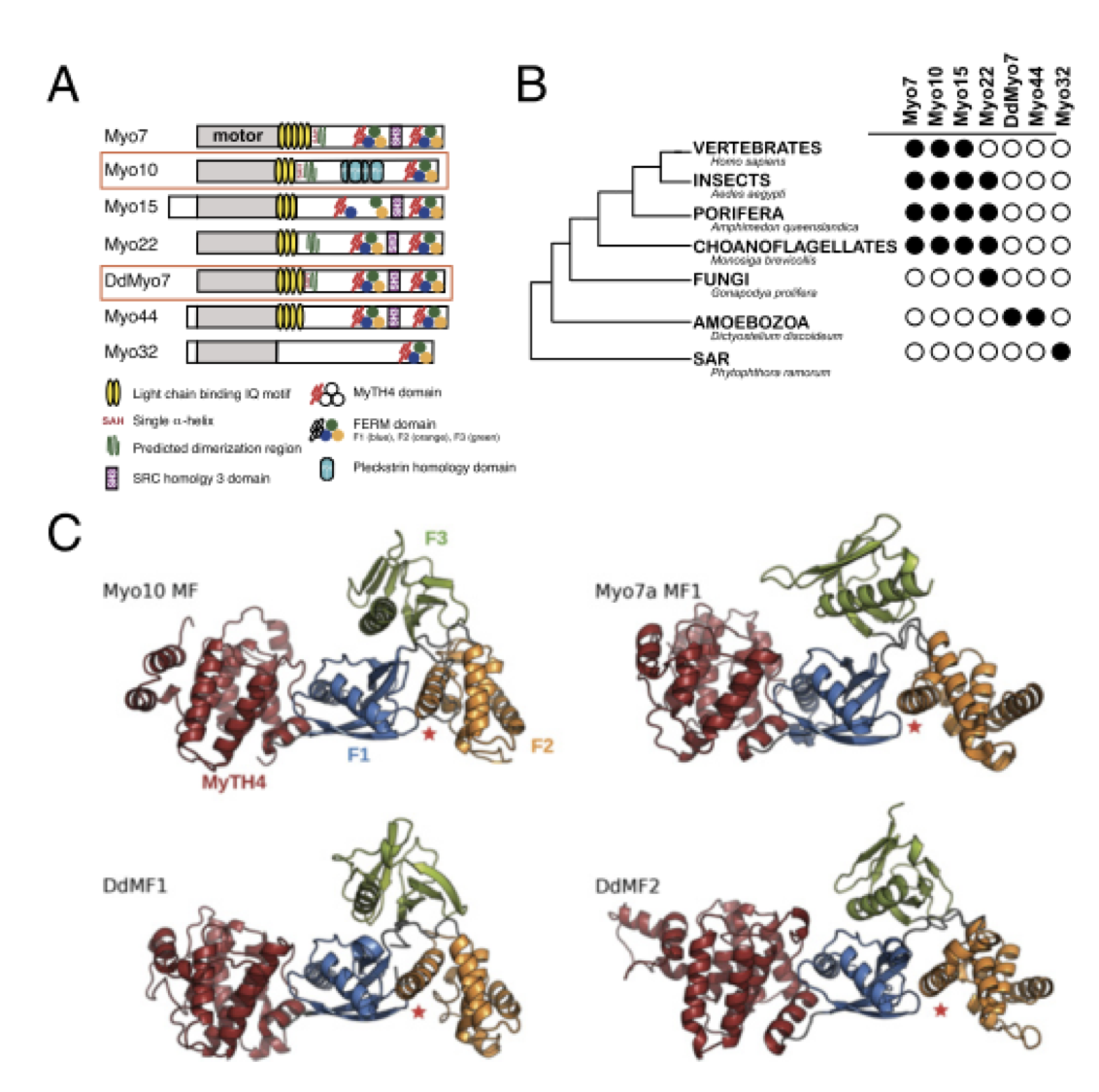
Myosin MyTH4-FERM structures highlight important principles of convergent evolution.
Planelles-Herrero VJ, Blanc F, Sirigu S, Sirkia H, Clause J, Sourigues Y, Johnsrud DO, Amigues B, Cecchini M, Gilbert SP, Houdusse A* & Titus MA* (2016) PNAS 113:E2906-2915. PDF Link
* co-corresponding authors
Abstract:
Myosins containing MyTH4-FERM (myosin tail homology 4-band 4.1, ezrin, radixin, moesin, or MF) domains in their tails are found in a wide range of phylogenetically divergent organisms, such as humans and the social amoeba Dictyostelium (Dd). Interestingly, evolutionarily distant MF myosins have similar roles in the extension of actin-filled membrane protrusions such as filopodia and bind to microtubules (MT), suggesting that the core functions of these MF myosins have been highly conserved over evolution. The structures of two DdMyo7 signature MF domains have been determined and comparison with mammalian MF structures reveals that characteristic features of MF domains are conserved. However, across millions of years of evolution conserved class-specific insertions are seen to alter the surfaces and the orientation of subdomains with respect to each other, likely resulting in new sites for binding partners. The MyTH4 domains of Myo10 and DdMyo7 bind to MT with micromolar affinity but, surprisingly, their MT binding sites are on opposite surfaces of the MyTH4 domain. The structural analysis in combination with comparison of diverse MF myosin sequences provides evidence that myosin tail domain features can be maintained without strict conservation of motifs. The results illustrate how tuning of existing features can give rise to new structures while preserving the general properties necessary for myosin tails. Thus, tinkering with the MF domain enables it to serve as a multifunctional platform for cooperative recruitment of various partners, allowing common properties such as autoinhibition of the motor and microtubule binding to arise through convergent evolution.
Selective localization of myosin-I proteins in macropinosomes and actin waves.
Brzeska H, Koech H, Pridham KJ, Korn ED & Titus MA (2016) Cytoskeleton 73:68-82. PDF Link
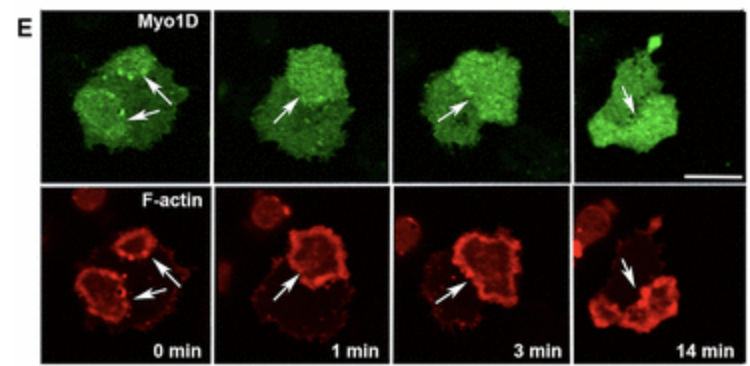
Class I myosins are widely expressed with roles in endocytosis and cell migration in a variety of cell types. Dictyostelium express multiple myosin Is, including three short-tailed (Myo1A, Myo1E, Myo1F) and three long-tailed (Myo1B, Myo1C, Myo1D). Here we report the molecular basis of the specific localizations of short-tailed Myo1A, Myo1E, and Myo1F compared to our previously determined localization of long-tailed Myo1B. Myo1A and Myo1B have common and unique localizations consistent with the various features of their tail region; specifically the BH sites in their tails are required for their association with the plasma membrane and heads are sufficient for relocalization to the front of polarized cells. Myo1A does not localize to actin waves and macropinocytic protrusions, in agreement with the absence of a tail region which is required for these localizations of Myo1B. However, in spite of the overall similarity of their domain structures, the cellular distributions of Myo1E and Myo1F are quite different from Myo1A. Myo1E and Myo1F, but not Myo1A, are associated with macropinocytic cups and actin waves. The localizations of Myo1E and Myo1F in macropinocytic structures and actin waves differ from the localization of Myo1B. Myo1B colocalizes with F-actin in the actin waves and at the tips of mature macropinocytic cups whereas Myo1E and Myo1F are in the interior of actin waves and along the entire surface of macropinocytic cups. Our results point to different mechanisms of targeting of short- and long-tailed myosin Is, and are consistent with these myosins having both shared and divergent cellular functions.
The association of Myosin IB with actin waves in Dictyostelium requires both the plasma membrane-binding site and actin-binding region in the myosin tail.
Brzeska H, Pridham K, Chery G, Titus MA & Korn ED (2014). PLoS One 9:e94306 PDF Link
Abstract:
F-actin structures and their distribution are important determinants of the dynamic shapes and functions of eukaryotic cells. Actin waves are F-actin formations that move along the ventral cell membrane driven by actin polymerization. Dictyostelium myosin IB is associated with actin waves but its role in the wave is unknown. Myosin IB is a monomeric, non-filamentous myosin with a globular head that binds to F-actin and has motor activity, and a non-helical tail comprising a basic region, a glycine-proline-glutamine-rich region and an SH3-domain. The basic region binds to acidic phospholipids in the plamsma membrane through a short basic-hydrophobic site in the basic region and the Gly-Pro-Gln region binds F-actin. In the current work we found that both the basic-hydrophobic site in the basic region and the Gly-Pro-Gln region of the tail are required for the association of myosin IB with actin waves. This is the first evidence that the Gly-Pro-Gln region is required for localization of myosin IB to a specific actin structure in situ. The head is not required for myosin IB association with actin waves but binding of the head to F-actin strengthens the association of myosin IB with waves and stabilizes waves. Neither the SH3-domain nor motor activity is required for association of myosin IB with actin waves. We conclude that myosin IB contributes to anchoring actin waves to the plasma membranes by binding of the basic hydrophobic site to acidic phospholipids in the plasma membrane and binding of the Gly-Pro-Gln region to F-actin in the wave.
Structural basis of Myosin V Rab GTPase-dependent cargo recognition.
Pylypenko O, Attanda W, Gauquelin C, Lahmani M, Coulibaly D, Baron B, Hoos S, Titus MA, England P & Houdusse AM (2013) PNAS 110:20443-20448. PDF Link
Abstract:
Specific recognition of the cargo that molecular motors transport or tether to cytoskeleton tracks allows them to perform precise cellular functions at particular times and positions in cells. However, very little is known about how evolution has favored conservation of functions for some isoforms, while also allowing for the generation of new recognition sites and specialized cellular functions. Here we present several crystal structures of the myosin Va or the myosin Vb globular tail domain (GTD) that gives insights into how the motor is linked to the recycling membrane compartments via Rab11 or to the melanosome membrane via recognition of the melanophilin adaptor that binds to Rab27a. The structures illustrate how the Rab11-binding site has been conserved during evolution and how divergence at another site of the GTD allows more specific interactions such as the specific recognition of melanophilin by the myosin Va isoform. With atomic structural insights, these structures also show how either the partner or the GTD structural plasticity upon association is critical for selective recruitment of the motor.
Allosteric communication in Dictyostelium Myosin II
Guhathakurta P, Prochniewicz E, Muretta JM, Titus MA & Thomas DD (2012) J Muscle Res Cell Motil 33:305-312. PDF Link
Abstract:
Myosin’s affinities for nucleotides and actin are reciprocal. Actin-binding substantially reduces the affinity of ATP for myosin, but the effect of actin on myosin’s ADP affinity is quite variable among myosin isoforms, serving as the principal mechanism for tuning the actomyosin system to specific physiological purposes. To understand the structural basis of this variable relationship between actin and ADP binding, we studied several constructs of the catalytic domain of Dictyostelium myosin II, varying their length (from the N-terminal origin) and cysteine content. The constructs varied considerably in their actin-activated ATPase activity and in the effect of actin on ADP affinity. Actin had no significant effect on ADP affinity for a single-cysteine catalytic domain construct, a double-cysteine construct partially restored the actin-dependence of ADP binding, and restoration of all native Cys restored it further, but full restoration of function (similar to that of skeletal muscle myosin II) was obtained only by adding all native Cys and an artificial lever arm extension. Pyrene-actin fluorescence confirmed these effects on ADP binding to actomyosin. We conclude that myosin’s Cys content and lever arm both allosterically modulate the reciprocal affinities of myosin for ADP and actin, a key determinant of the biological functions of myosin isoforms.
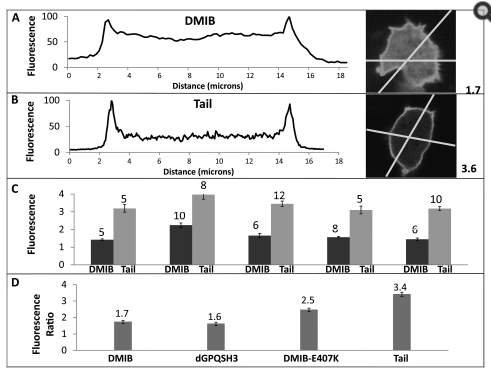
Molecular basis of dynamic relocalization of Dictyostelium Myosin IB.
Brzeska H, Guag J, Preston GM, Titus MA & Korn ED (2012) J Biol Chem 287:14923-14936. PDF Link
Abstract:
Class I myosins have a single heavy chain co
mprising an N-terminal motor domain with actin-activated ATPase activity and a C-terminal globular tail with a basic region that binds to acidic phospholipids. These myosins contribute to the formation of actin-rich protrusions such as pseudopodia, but regulation of the dynamic localization to these structures is not understood. Previously, we found that Acanthamoebain vitro myosin IC binds to acidic phospholipids through a short sequence of basic and hydrophobic amino acids, BH site, based on the charge density of the phospholipids. The tail of Dictyostelium in vivo myosin IB (DMIB) also contains a BH site. We now report that the BH site is essential for DMIB binding to the plasma membrane and describe the molecular basis of the dynamic relocalization of DMIB in live cells. Endogenous DMIB is localized uniformly on the plasma membrane of resting cells, at active protrusions and cell-cell contacts of randomly moving cells, and at the front of motile polarized cells. The BH site is required for association of DMIB with the plasma membrane at all stages where it colocalizes with phosphoinositide bisphosphate/phosphoinositide trisphosphate (PIP2/PIP3). The charge-based specificity of the BH site allows for specificity of DMIB for PIP2/PIP3 similar to the PH domain-based specificity of other class I myosins. However, DMIB-head is required for relocalization of DMIB to the front of migrating cells. Motor activity is not essential, but the actin binding site in the head is important. Thus, dynamic relocalization of DMIB is determined principally by the local PIP2/PIP3 concentration in the plasma membrane and cytoplasmic F-actin.
Structural and functional impact of site-directed methionine oxidation in myosin.
Klein JC, Moen RJ, Smith EA, Titus MA & Thomas DD (2011) Biochemistry 50:10318-10327. PDF Link
Abstract:
We have examined the structural and functional effects of site-directed methionine oxidation in Dictyostelium (Dicty) myosin II using mutagenesis, in vitro oxidation, and site-directed spin-labeling for electron paramagnetic resonance (EPR). Protein oxidation by reactive oxygen and nitrogen species is critical for normal cellular function, but oxidative stress has been implicated in disease progression and biological aging. Our goal is to bridge understanding of protein oxidation and muscle dysfunction with molecular-level insights into actomyosin interaction. In order to focus on methionine oxidation and to facilitate site-directed spectroscopy, we started with a Cys-lite version of Dicty myosin II. For Dicty myosin containing native methionines, peroxide treatment decreased actin-activated myosin ATPase activity, consistent with the decline in actomyosin function previously observed in biologically aged or peroxide-treated muscle. Methionine-to-leucine mutations, used to protect specific sites from oxidation, identified a single methionine that is functionally sensitive to oxidation: M394, near the myosin cardiomyopathy loop in the actin-binding interface. Previously characterized myosin labeling sites for spectroscopy in the force-producing region and actin-binding cleft were examined; spin-label mobility and distance measurements in the actin-binding cleft were sensitive to oxidation, but particularly in the presence of actin. Overall secondary structure and thermal stability were unaffected by oxidation. We conclude that the oxidation-induced structural change in myosin includes a redistribution of existing structural states of the actin-binding cleft. These results will be applicable to the many biological and therapeutic contexts in which a detailed understanding of protein oxidation as well as function and structure relationships is sought.
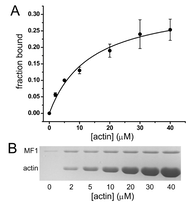
Characterization of a Myosin VII MyTH/FERM domain.
Moen RJ, Johnsrud DO, Thomas DD & Titus MA. (2011) J Mol Biol 413:17-23 PDF Link
Abstract:
A group of closely related myosins is characterized by the presence of at least one MyTH/FERM (myosin tail homology; band 4.1, ezrin, radixin, moesin) domain in their C-terminal tails. This domain interacts with a variety of binding partners, and mutations in either the MyTH4 or the FERM domain of myosin VII and myosin XV result in deafness, highlighting the functional importance of each domain. The N-terminal MyTH/FERM region of Dictyostelium myosin VII (M7) has been isolated as a first step toward gaining insight into the function of this domain and its interaction with binding partners. The M7 MyTH4/FERM domain (MF1) binds to both actin and microtubules in vitro, with dissociation constants of 13.7 and 1.7 μM, respectively. Gel filtration and UV spectroscopy reveal that MF1 exists as a monomer in solution and forms a well-folded, compact conformation with a high degree of secondary structure. These results indicate that MF1 forms an integrated structural domain that serves to couple actin filaments and microtubules in specific regions of the cytoskeleton.
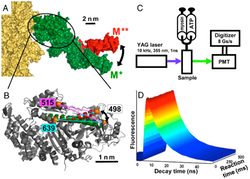
Structural Kinetics of Myosin By Transient Time-Resolved FRET.
Nesmelov YE, Agafonov RV, Negrashov IV, Blakely SB, Titus MA & Thomas DD (2010) PNAS 108:1891-1896. PDF Link
Abstract:
For many proteins, especially for molecular motors and other enzymes, the functional mechanisms remain unsolved due to a gap between static structural data and kinetics. We have filled this gap by detecting structure and kinetics simultaneously. This structural kinetics experiment is made possible by a new technique, (TR)2FRET (transient time-resolved FRET), which resolves protein structural states on the submillisecond timescale during the transient phase of a biochemical reaction. (TR)2FRET is accomplished with a fluorescence instrument that uses a pulsed laser and direct waveform recording to acquire an accurate subnanosecond time-resolved fluorescence decay every 0.1 ms after stopped flow. To apply this method to myosin, we labeled the force-generating region site specifically with two probes, mixed rapidly with ATP to initiate the recovery stroke, and measured the interprobe distance by (TR)2FRET with high resolution in both space and time. We found that the relay helix bends during the recovery stroke, most of which occurs before ATP is hydrolyzed, and two structural states (relay helix straight and bent) are resolved in each nucleotide-bound biochemical state. Thus the structural transition of the force-generating region of myosin is only loosely coupled to the ATPase reaction, with conformational selection driving the motor mechanism.
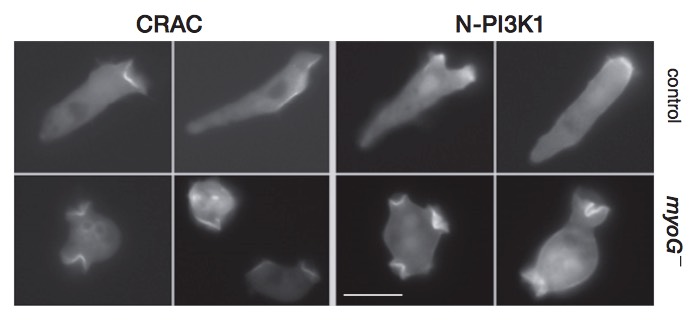
An unconventional myosin required for cell polarization and chemotaxis.
Breshears LM, Wessels D, Soll DR & Titus MA (2010) PNAS 107:6918-6923. PDF Link
Abstract:
MyTH/FERM (myosin tail homology 4/band 4.1, ezrin, radixin, and moesin) myosins have roles in cellular adhesion, extension of actin-filled projections such as filopodia and stereocilia, and directional migration. The amoeba Dictyostelium discoideum expresses a simple complement of MyTH/FERM myosins, a class VII (M7) myosin required for cell-substrate adhesion and a unique myosin named MyoG. Mutants lacking MyoG exhibit a wide range of normal actin-based behaviors, including chemotaxis to folic acid, but have a striking defect in polarization and chemotaxis to cAMP. Although the myoG mutants respond to cAMP stimulation by increasing persistence and weakly increasing levels of cortical F-actin, they do not polarize; instead, they maintain a round shape and move slowly and randomly when exposed to a chemotactic gradient. The mutants also fail to activate and localize PI3K to the membrane closest to the source of chemoattractant. These data reveal a role for a MyTH/FERM myosin in mediating early chemotactic signaling and suggest that MyTH/FERM proteins have conserved roles in signaling and the generation of cell polarity.
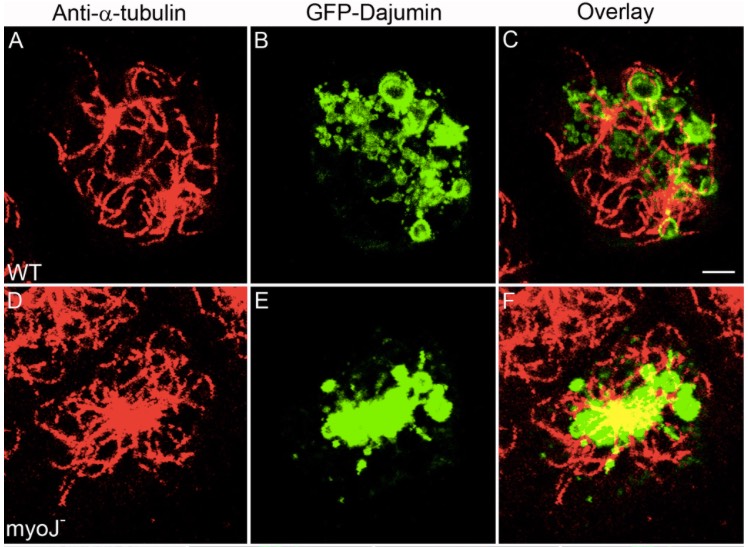
The Dictyostelium type V myosin MyoJ is responsible for the cortical association and motility of contractile vacuole membranes.
Jung G, Titus MA & Hammer JA 3rd (2009) J Cell Biol 186:555-570. PDF Link
Abstract:
The contractile vacuole (CV) complex in Dictyostelium is a tubulovesicular osmoregulatory organelle that exhibits extensive motility along the actin-rich cortex, providing a useful model for investigating myosin-dependent membrane transport. Here, we show that the type V myosin myoJ localizes to CV membranes and is required for efficient osmoregulation, the normal accumulation of CV membranes in the cortex, and the conversion of collapsed bladder membranes into outwardly radiating cortical CV tubules. Complementation of myoJ-null cells with a version of myoJ containing a shorter lever arm causes these radiating tubules to move at a slower speed, confirming myoJ's role in translocating CV membranes along the cortex. MyoJ-null cells also exhibit a dramatic concentration of CV membranes around the microtubule-organizing center. Consistently, we demonstrate that CV membranes also move bi-directionally on microtubules between the cortex and the centrosome. Therefore, myoJ cooperates with plus and minus end-directed microtubule motors to drive the normal distribution and dynamics of the CV complex in Dictyostelium.
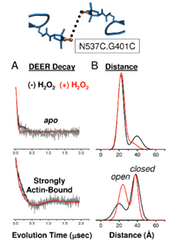
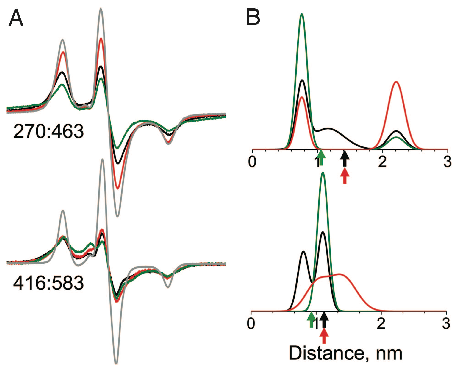
Actin-binding Cleft Closure In Myosin II Probed By Site-Directed Spin Labeling And Pulsed EPR.
Klein JC, Burr AR, Svensson B, Kennedy DJ, Titus MA, Rayment I, Thomas DD. (2008). P.N.A.S. 150:12867-12872 PDF Link
Abstract:
We present a structurally dynamic model for nucleotide- and actin-induced closure of the actin-binding cleft of myosin, based on site-directed spin labeling and electron paramagnetic resonance (EPR) in Dictyostelium myosin II. The actin-binding cleft is a solvent-filled cavity that extends to the nucleotide-binding pocket and has been predicted to close upon strong actin binding. Single-cysteine labeling sites were engineered to probe mobility and accessibility within the cleft. Addition of ADP and vanadate, which traps the posthydrolysis biochemical state, influenced probe mobility and accessibility slightly, whereas actin binding caused more dramatic changes in accessibility, consistent with cleft closure. We engineered five pairs of cysteine labeling sites to straddle the cleft, each pair having one label on the upper 50-kDa domain and one on the lower 50-kDa domain. Distances between spin-labeled sites were determined from the resulting spin–spin interactions, as measured by continuous wave EPR for distances of 0.7–2 nm or pulsed EPR (double electron–electron resonance) for distances of 1.7–6 nm. Because of the high distance resolution of EPR, at least two distinct structural states of the cleft were resolved. Each of the biochemical states tested (prehydrolysis, posthydrolysis, and rigor), reflects a mixture of these structural states, indicating that the coupling between biochemical and structural states is not rigid. The resulting model is much more dynamic than previously envisioned, with both open and closed conformations of the cleft interconverting, even in the rigor actomyosin complex.